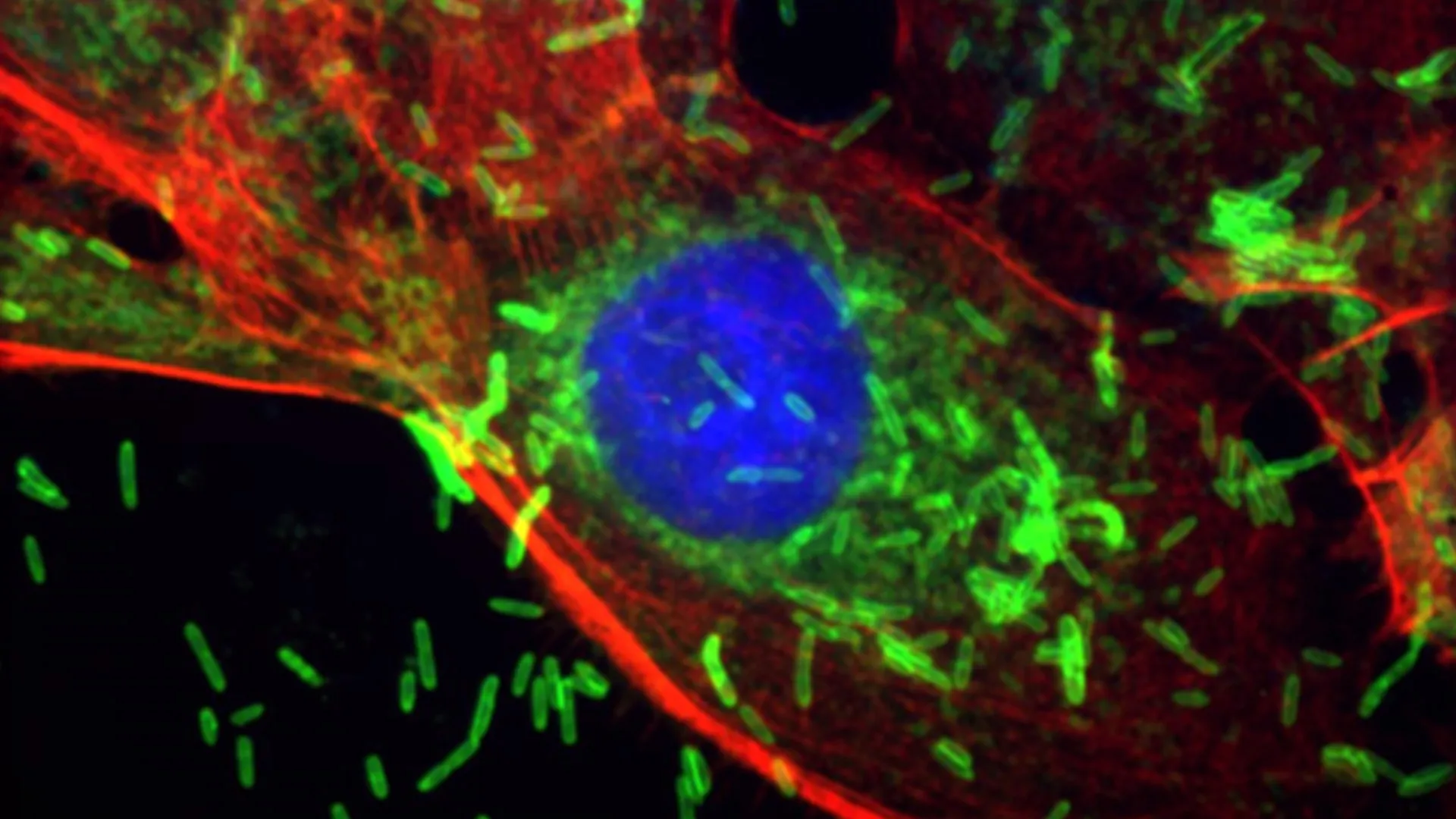
Published in the esteemed journal Cell Communication and Signaling, the study meticulously details how Fusobacterium nucleatum (F. nucleatum), a microbe previously implicated in colorectal and other cancers, can transcend its typical oral habitat. The researchers demonstrated that this bacterium possesses the remarkable ability to enter the bloodstream from inflamed oral tissues and subsequently colonize breast tissue. Once established in the breast, F. nucleatum acts as a potent trigger, initiating a cascade of inflammatory responses and early cellular changes that are hallmarks of cancer development. Through rigorous animal models designed to mimic human breast cancer, the presence of F. nucleatum was unequivocally shown to dramatically expedite primary tumor growth and significantly enhance the dissemination of cancer cells from the breast to distant organs, most notably the lungs, a critical step in advanced disease.
"The key takeaway is that this oral microbe can reside in breast tissue and that there is a connection between this pathogen and breast cancer," stated Dr. Cynthia Sharma, a leading researcher at Johns Hopkins and a senior author of the study. Dr. Sharma emphasized that the team’s initial inspiration for this deep dive into the underlying mechanisms stemmed from a growing body of epidemiological evidence. Numerous smaller studies, collectively analyzing data from thousands of patients, had consistently reported a statistical association between periodontal disease—a chronic inflammatory condition of the gums—and an elevated risk of breast cancer. "We wanted to dig deeper and see if we could uncover the underlying connections," added Dr. Sheetal Parida, the first author of the study and a dedicated research associate working alongside Dr. Sharma. This collective motivation pushed the research beyond mere correlation, aiming to establish a causative link and elucidate the precise biological pathways involved.
Understanding Fusobacterium nucleatum and Its Pathogenic Potential
To fully appreciate the implications of these findings, it’s crucial to understand Fusobacterium nucleatum. It is a ubiquitous gram-negative anaerobic bacterium, meaning it thrives in oxygen-deprived environments, making the crevices of the oral cavity an ideal habitat. F. nucleatum is a prominent member of the oral microbiome, particularly known for its role in forming dental plaque and biofilm. While present in healthy mouths, its overgrowth and dysregulation are strongly associated with periodontal disease, also known as gum disease.
Periodontal disease encompasses a spectrum of conditions, from mild gingivitis (gum inflammation) to severe periodontitis, which involves the destruction of the soft tissue and bone supporting the teeth. It affects a significant portion of the global population, with severe forms impacting millions worldwide. The hallmark of periodontal disease is chronic inflammation, which creates an environment ripe for bacterial invasion and systemic dissemination. The inflamed and bleeding gums provide a gateway for oral bacteria, including F. nucleatum, to enter the bloodstream—a phenomenon known as transient bacteremia.
The concept of oral bacteria influencing distant sites is not entirely new. F. nucleatum, in particular, has gained notoriety for its involvement in colorectal cancer (CRC). Research has shown that F. nucleatum can colonize colorectal tumors, where it creates an inflammatory microenvironment that promotes tumor growth, suppresses anti-tumor immune responses, and contributes to chemotherapy resistance. Its presence in CRC tumors is often linked to more aggressive disease and poorer patient outcomes. This established precedent for F. nucleatum’s oncogenic role in the gut provided a compelling rationale for investigating its potential involvement in other cancers, including breast cancer. The current study significantly expands this understanding by demonstrating its ability to specifically target and impact breast tissue.
Unveiling the Mechanisms: From Colonization to Malignant Transformation
The Johns Hopkins team employed a sophisticated array of experimental models to dissect how F. nucleatum exerts its influence. Their experiments using both in vivo mouse models and in vitro human breast cancer cells provided a comprehensive picture of the bacterium’s impact.
When F. nucleatum was introduced directly into the mammary ducts of mice, mimicking a localized infection, the researchers observed profound cellular alterations. These included the development of metaplastic and hyperplastic lesions—non-cancerous changes characterized by excessive cell growth or a shift in cell type. While not cancerous themselves, these lesions are recognized as pre-malignant conditions, indicating an increased risk for future malignant transformation. Crucially, these cellular changes were accompanied by robust inflammation, a well-established driver of carcinogenesis. Furthermore, direct exposure to F. nucleatum led to increased cell proliferation and, significantly, direct damage to cellular DNA, two fundamental steps in the initiation of cancer.
Perhaps even more alarming were the findings when the bacterium entered the bloodstream, simulating a systemic spread from an oral infection. In these scenarios, F. nucleatum profoundly boosted the growth of existing tumors and, critically, amplified the metastatic potential of cancer cells. Metastasis, the spread of cancer from its primary site to other parts of the body, is responsible for the vast majority of cancer-related deaths, making any factor that promotes it a significant concern. The ability of an oral microbe to accelerate this deadly process underscores the gravity of the research.
DNA Damage and Tumor-Promoting Changes: The Molecular Underpinnings
The team’s research further delved into the precise molecular mechanisms by which F. nucleatum wreaks havoc. They uncovered a key biological process linking bacterial presence to aggressive cancer phenotypes. Exposure to F. nucleatum was found to induce significant damage to cellular DNA, a constant threat that cells typically manage through sophisticated repair systems. However, F. nucleatum’s influence extended to activating DNA repair systems that are notably prone to errors.
One such pathway identified was non-homologous end joining (NHEJ). NHEJ is a rapid "quick fix" mechanism for repairing double-strand breaks in DNA, which are among the most dangerous forms of DNA damage. While efficient, NHEJ is notorious for its error-prone nature. Unlike homologous recombination, a more precise repair pathway that uses an intact template, NHEJ simply ligates broken DNA ends together, often leading to insertions, deletions, or translocations. These genomic alterations can introduce mutations that activate oncogenes or inactivate tumor suppressor genes, thereby driving cancer development and progression.
Even short-term exposure to F. nucleatum was sufficient to elevate levels of a specific protein called PKcs (DNA-dependent protein kinase catalytic subunit). PKcs is a crucial component of the NHEJ pathway. The researchers demonstrated a direct link between increased PKcs levels and a more aggressive cancer cell phenotype. Specifically, elevated PKcs was associated with:
- Greater cancer cell movement and invasion: This enhances the ability of cancer cells to break away from the primary tumor and infiltrate surrounding tissues, a prerequisite for metastasis.
- Stem-like traits: Cancer stem cells are a subpopulation of cancer cells with self-renewal capacity, resistance to conventional therapies, and a heightened ability to initiate new tumors. Promoting stem-like traits makes cancer more aggressive and harder to treat.
- Resistance to chemotherapy: A major clinical challenge in cancer treatment, as cells that are resistant to chemotherapy are more likely to survive treatment and cause relapse.
These findings provide a compelling molecular explanation for how F. nucleatum can not only initiate cancer but also drive its progression and contribute to therapeutic resistance, fundamentally altering the biological behavior of breast cancer cells.
Higher Risk in Genetically Susceptible Individuals: The BRCA1 Connection
The study also shed light on why certain individuals might be more susceptible to the oncogenic effects of F. nucleatum, identifying a critical interaction with genetic predisposition. Epithelial cells, which line the breast ducts where most breast cancers originate, and particularly breast cancer cells carrying mutations in the BRCA1 gene, appeared to be especially vulnerable.
The BRCA1 gene is a well-known tumor suppressor gene. Mutations in BRCA1 significantly increase a woman’s lifetime risk of developing breast and ovarian cancer. BRCA1 plays a crucial role in DNA repair, specifically in the high-fidelity homologous recombination pathway. When BRCA1 is mutated, this precise repair mechanism is compromised, making cells more reliant on error-prone pathways like NHEJ or simply more prone to accumulating DNA damage.
The researchers discovered a fascinating mechanism behind the heightened susceptibility of BRCA1-mutant cells. These cells exhibited elevated levels of a specific surface sugar, Gal-GalNAc. This sugar acts as a "receptor" or attachment point, facilitating the binding and entry of F. nucleatum into the cells. Consequently, BRCA1-mutant cells absorbed more F. nucleatum and, critically, retained the bacterium over extended periods, even across multiple cell generations. This persistent intracellular presence of F. nucleatum in already vulnerable cells intensified DNA damage and amplified the cancer-promoting effects, creating a dangerous synergy between a genetic predisposition and an environmental microbial factor.
"Our findings reveal a link between oral microbes and breast cancer risk and progression, particularly in genetically susceptible individuals," emphasized Dr. Sharma. This statement underscores a paradigm shift in understanding cancer etiology. "Nothing happens in isolation. The results suggest that multiple risk factors come together with F. nucleatum acting as an environmental factor that may cooperate with inherited BRCA1 mutations to promote breast cancer and tumor aggressiveness." This concept of a complex interplay between genetics, environment, and the microbiome opens new avenues for personalized risk assessment and targeted interventions.
Implications for Oral Health, Cancer Risk, and Future Directions
The profound implications of this research extend across multiple domains, from public health to personalized cancer therapy. While the researchers rightly emphasize that more work is needed to fully translate these findings into patient care, the study lays a robust foundation for future clinical applications.
For Public Health and Prevention: The most immediate and accessible implication is the potential for maintaining excellent oral health to play a role in reducing breast cancer risk. Periodontal disease is largely preventable and treatable through good oral hygiene practices (brushing, flossing) and regular dental check-ups. If F. nucleatum from the oral cavity is indeed a causative factor in breast cancer, then improving oral health could represent a novel, modifiable risk factor. This could be particularly significant for individuals with a family history of breast cancer or those carrying BRCA1 mutations, who are already at elevated risk. Public health campaigns promoting oral hygiene could gain a new, compelling dimension.
For Cancer Screening and Diagnosis: The discovery raises the possibility that F. nucleatum could serve as a biomarker. Could the presence or abundance of F. nucleatum in saliva, blood, or even breast tissue samples be an indicator of increased breast cancer risk or a predictor of tumor aggressiveness? Future diagnostic tools might incorporate microbial profiling to identify at-risk individuals or guide treatment strategies.
For Therapeutic Interventions: If F. nucleatum actively drives breast cancer progression, then targeting the bacterium itself could become a novel therapeutic strategy. This could involve:
- Antibiotics: Specific antimicrobial agents to eradicate F. nucleatum in the oral cavity or within breast tumors.
- Probiotics or prebiotics: Modulating the oral and gut microbiomes to suppress F. nucleatum or promote beneficial bacteria.
- Vaccines: Developing vaccines against F. nucleatum, similar to those against other pathogenic bacteria, could offer a long-term preventive solution.
- Targeting bacterial adhesion: Developing drugs that block the Gal-GalNAc receptor or other adhesion mechanisms could prevent F. nucleatum from colonizing breast tissue, particularly in high-risk individuals like those with BRCA1 mutations.
- Targeting host response: Interventions aimed at mitigating the inflammatory response or DNA damage pathways triggered by F. nucleatum could also be explored.
Future Research Avenues: The Johns Hopkins team has opened a fascinating new frontier, and the next steps are clear. Large-scale human epidemiological studies are essential to confirm these findings in diverse patient populations. These studies would aim to establish a stronger causal link, quantify the risk, and identify specific subgroups most affected. Further mechanistic research is also needed to fully elucidate all the molecular pathways involved, explore the potential role of other oral or gut microbes in breast cancer, and investigate the interplay of the entire breast microbiome (a relatively new concept) with F. nucleatum. Ultimately, clinical trials testing interventions such as aggressive periodontal treatment or targeted antimicrobial therapies in high-risk individuals or breast cancer patients will be crucial to translate these discoveries into tangible improvements in patient outcomes.
In addition to Dr. Sharma and Dr. Parida, the dedicated research team included Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson, and Cynthia Sears, whose collective expertise made this seminal research possible. The study received vital financial backing from several prestigious organizations, including the Breast Cancer Research Foundation, Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program (grants BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention, and the Bloomberg~Kimmel Institute for Cancer Immunotherapy. Their support underscores the profound potential of this work to redefine our understanding of breast cancer and pave the way for innovative strategies in prevention, diagnosis, and treatment. This research from Johns Hopkins stands as a powerful testament to the evolving understanding of cancer, highlighting the intricate connections between distant biological systems and underscoring the vital role of our microbial inhabitants in shaping human health and disease.